The Michele Jacob Lab
APC Function in Cognitive Disorders
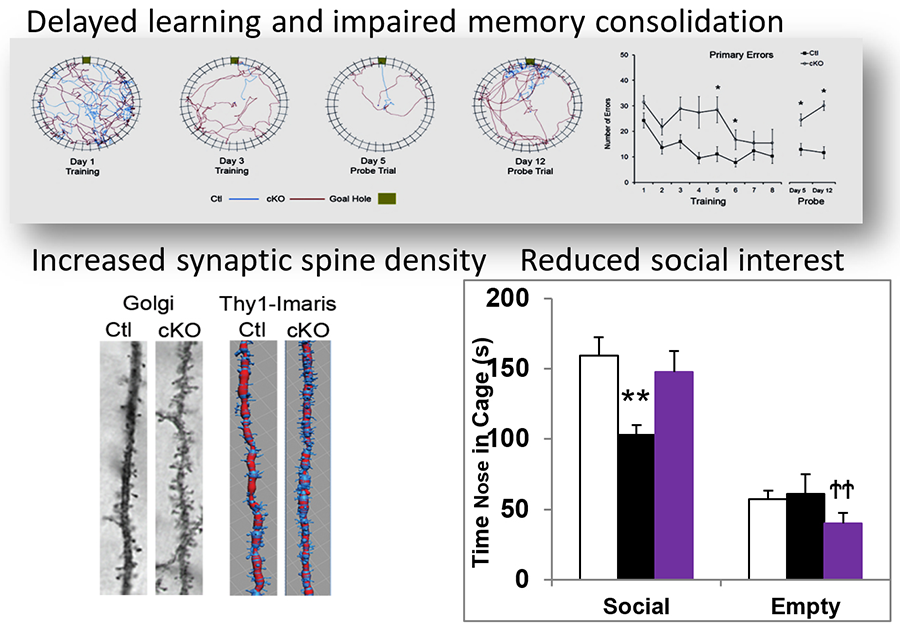
Several human gene mutations that link to intellectual disability and autism are predicted to cause malfunction of the adenomatous polyposis coli protein (APC), β-catenin (β-cat), canonical Wnt signaling pathway. To gain insights into the underlying pathophysiological changes, we have generated an APC conditional knock-out (cKO) mouse with APC deletion targeted to neurons during synaptic differentiation. APC cKO mice, compared with wild-type littermates, exhibit learning impairments, autism-like behaviors and chronic seizures (pediatric epileptic encephalopathies) (Mohn et al, 2014; Pirone et al, 2017). APC loss also leads to aberrant synaptic density, function, and plasticity. Additionally, APC cKOs display excessive β-catenin levels and associated changes in synaptic adhesion and signaling networks. These findings define a novel molecular etiology of overlapping cognitive impairments, autistic disabilities and chronic seizures.
Our current work is defining the changes downstream of APC that lead to these disorders. In particular, we are testing whether aberrant β-catenin/ Wnt signaling levels play a central role. We have generated novel β-catenin mutant mouse models with down- or up-regulation of this pathway. We are also testing whether pharmacological treatments can correct malfunction of this pathway and thereby amend the cognitive impairments, autistic disabilities and seizures in our mutant mouse models. Our findings are elucidating molecular, functional frameworks that link to learning deficits of different severities. Our drug treatment studies will provide proof-of-concept for new, effective strategies to ameliorate these disorders.
Figure 1. Malfunction of APC, β-catenin/ canonical Wnt pathways in neurons leads to increased synaptic density, cognitive impairments and autism-like behaviors.